The reaction between lead and hydrochloric acid
The chloride produced in the reaction between lead and hydrochloric acid is lead (II) chloride.
| Lead | + | Hydrochloric acid | → | Lead (II) chloride | + | Hydrogen |
| Pb | + | 2HCl | → | PbCl2 | + | H2 |
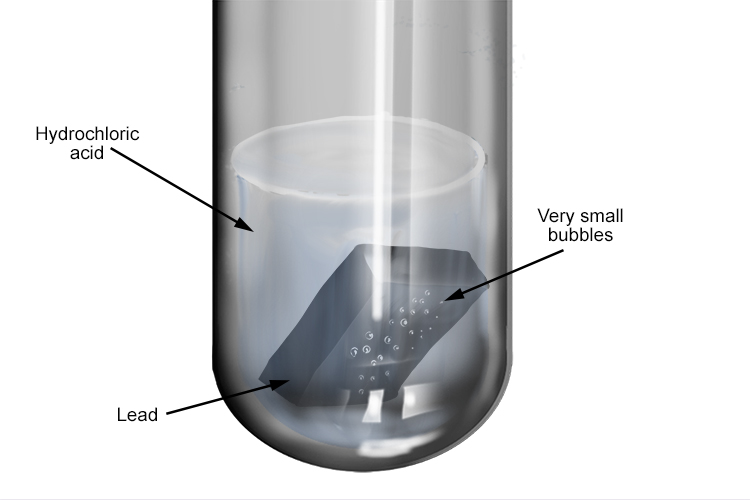
The reaction between hydrochloric acid and lead is very slow, producing only a few small bubbles of hydrogen gas. Lead is not used as a protective coating on other metals as it is poisonous.




