Naming hydrocarbons
We are going to teach you how to reproduce all of this and it’s easy.
Methane CH4
.jpg)
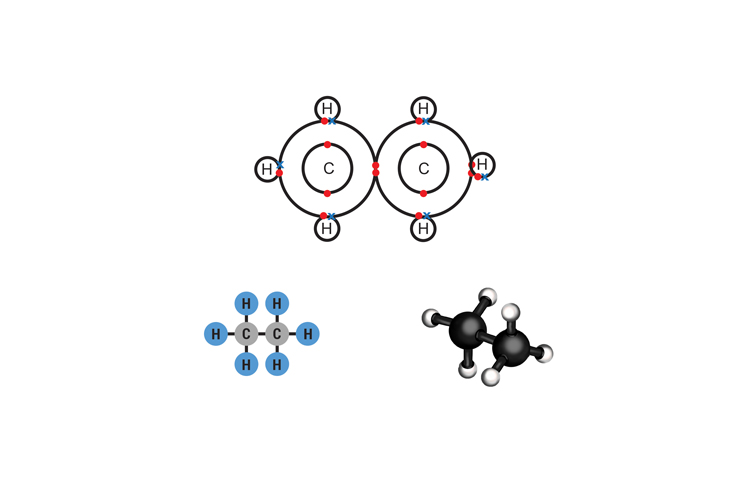
Ethane C2H6
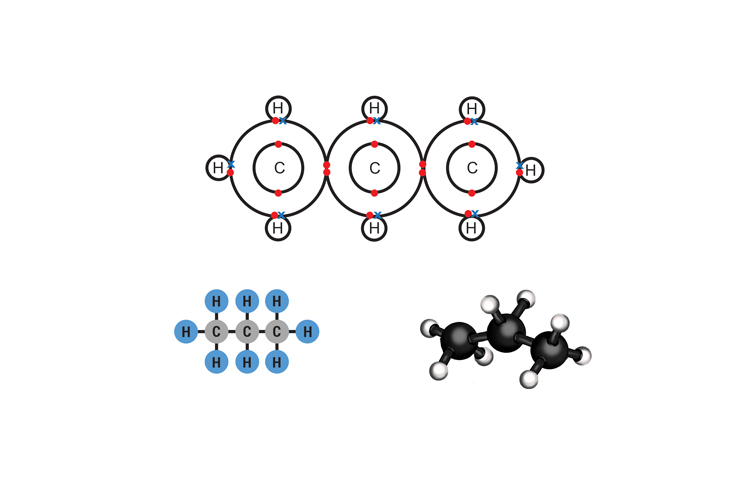
Propane C3H8
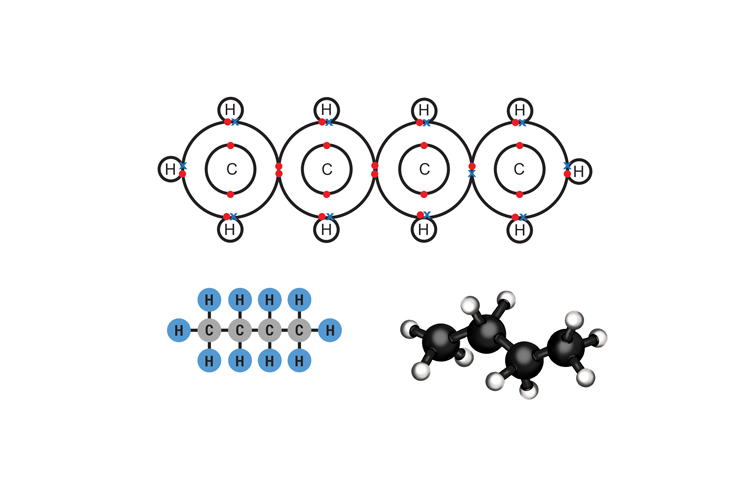
Butane C4H10
Methene
There is no such thing as methene
Because Methene would only have one carbon atom and a double carbon bond, it cannot exist.
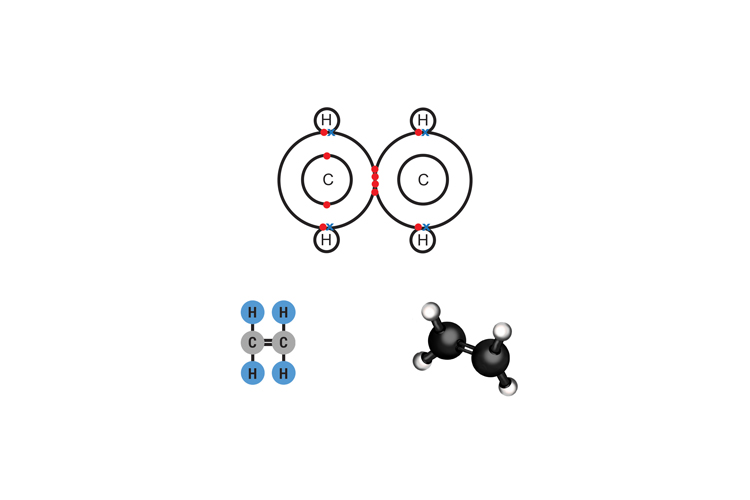
Ethene (sometimes called ethylene) C2H4
Propene C3H6
.jpg)
Butene (sometimes known as butylene) C4H8
.jpg)
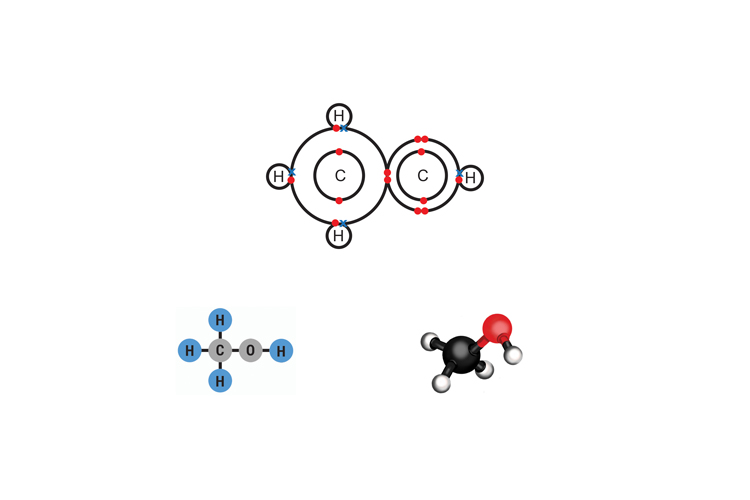
Methanol CH3OH
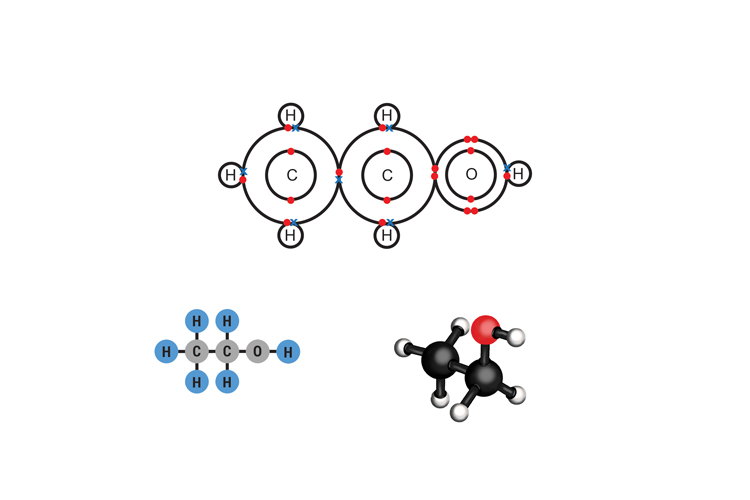
Ethanol C2H5OH
(Remember ethanol: alcohol and the drunk poodle.)
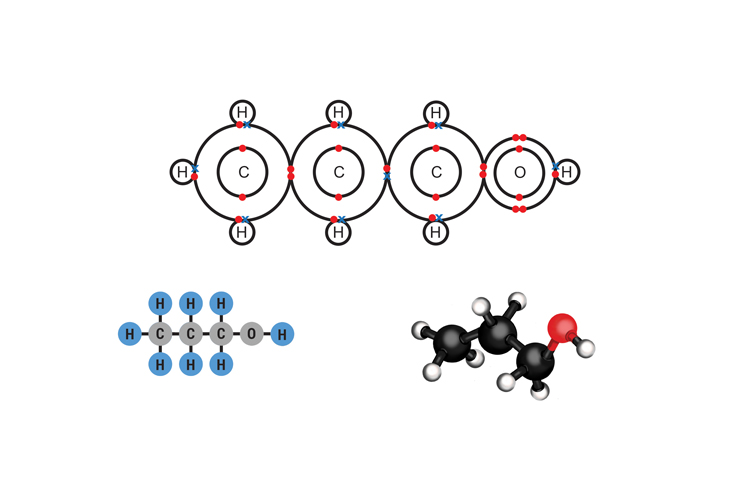
Propanol C3H7OH
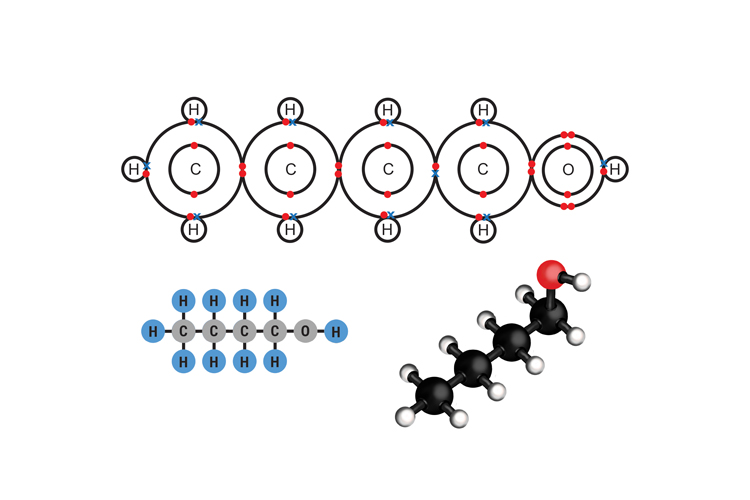
Butanol C4H9OH