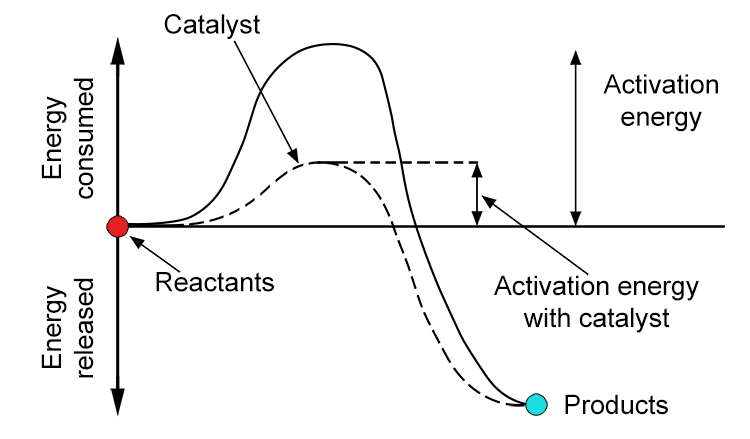
activation energy – the minimum energy required for a chemical reaction to occur
Note: Every chemical reaction requires a minimum energy input, called the activation energy to break existing bonds and start forming new ones.
To remember the meaning of activation energy, use the following mnemonic:
Energy was required to activate (activation energy) the dominos and begin the chain reaction.

To begin a chemical reaction, a minimum amount of energy is required for the reacting particles to collide successfully and start a reaction. Even if a reaction releases energy overall (exothermic reactions), the particles first need enough energy to break the existing bonds in the reactants before new bonds can form in the products. An example is needing a match to light wood.
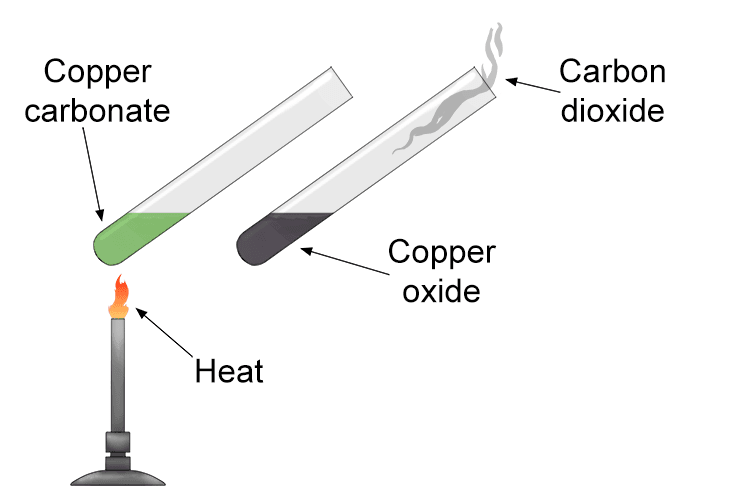
Think of activation energy as an energy barrier that must be overcome for a reaction to occur. This is why some reactions need heating to get started, as the heat provides the particles with enough kinetic energy to reach the activation energy. For example, copper carbonate decomposes and produces copper oxide and carbon dioxide when subjected to sustained heat.
Even rusting requires an activation energy. Imagine a tiny barrier that keeps iron safe from turning into rust instantly. That barrier is what scientists call activation energy.
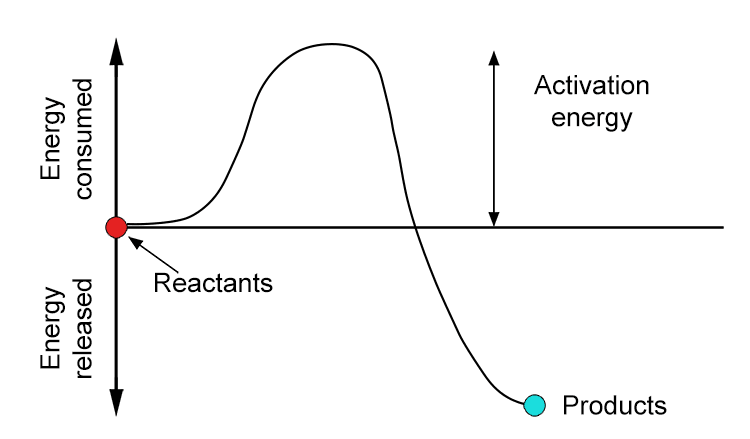
It is the smallest amount of energy needed for a chemical reaction to get started. When iron reacts with oxygen in the air to form rust, this energy barrier must be crossed first even though rusting is a process that releases energy overall it does not happen right away because the molecules involved need to gain enough energy to break their current bonds and form new ones. At room temperature molecules typically don't have enough energy to cross this barrier. Water and electrolytes are substances that can conduct electricity can make this process faster by lowering the activation energy.
Catalysts can be used to lower the activation energy required, making it easier for particles to react and therefore speeding up the reaction without being used up themselves. Reaction with high activation energies tend to be slower because fewer particles have sufficient energy to react at any given time.