conductor (chemistry) – a material which contains charged particles which are free to move to carry energy
(pronounced kuhn-duk-tuh)
To remember the meaning of conductor (chemistry), use the following mnemonic:
The conductor (conductor) charged his phone on the move with an energy pack he carried.
-in-chemistry-vocab-copy.820a88e.png)
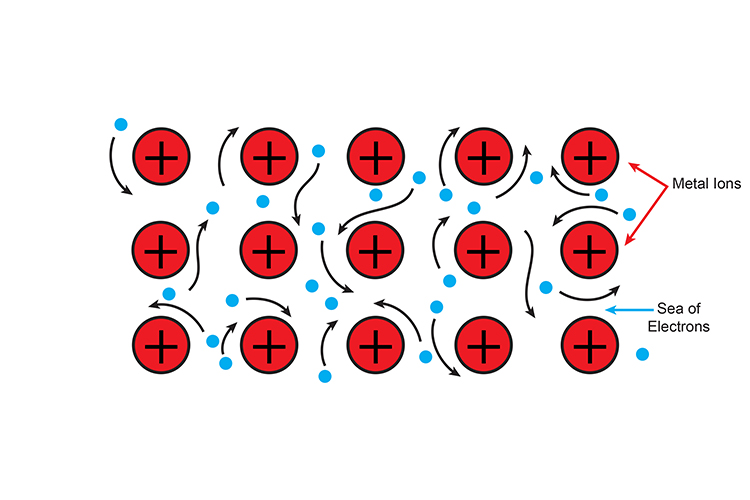
Conductors allow the flow of electrical charge (electricity) or thermal energy (heat) through it easily due to the presence of mobile charged particles, typically electrons. Electrical conductors, such as metals like copper, silver, aluminium, and gold, have structures where electrons are not tightly bound to individual atoms but instead exist in a "sea" of electrons that can move freely throughout the material when a voltage is applied, allowing electrical current to flow with minimal resistance.
Most metals are excellent conductors because their metallic bonding creates this free-moving electron structure, which is why metals are widely used in electrical wiring, circuits and electronic devices. Thermal conductors similarly allow heat energy to transfer through them efficiently, with metals again being excellent thermal conductors.