reversible reaction – a chemical reaction that proceeds in both forward and reverse directions simultaneously
To remember the meaning of reversible reaction, use the following mnemonic:
This lorry is reversible and gets a great reaction (reversible reaction) when it stops and goes forward and reverses.

Unlike irreversible reactions that only go in one direction until completion, reversible reactions can proceed in both the forward direction (reactants to products) and the backward direction (products to reactants) at the same time.
Rechargeable batteries are reversible because they use chemical reactions that can chemically run forwards (discharging to produce power) and reverse chemically (charging by applying external current) to restore their original materials allowing for repeated use.

Another reversable reaction can be seen when experimenting with copper sulphate:
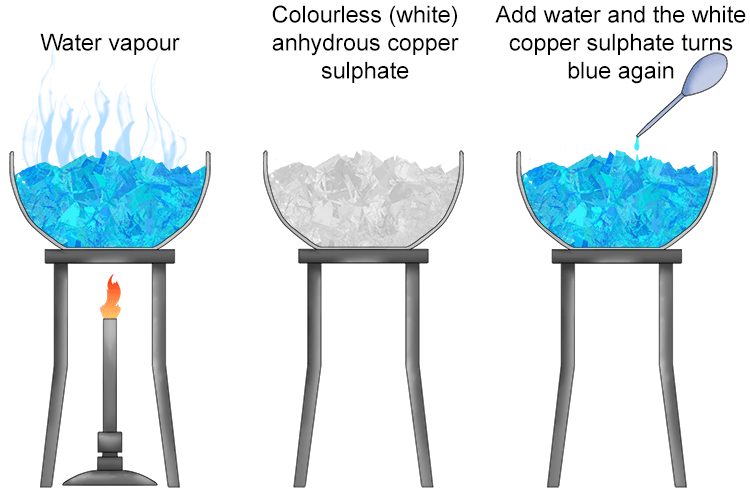
If you heat up hydrated copper sulphate crystals the water will evaporate off.
After heating you are left with anhydrous copper sulphate. This is colourless/white solid.
Allow the anhydrous copper sulphate to cool and then add water, you then get back to the hydrated blue copper sulphate again.
Copper sulphate is a colourless/white ionic crystalline solid. If you have a bottle of copper sulphate in the lab it will be blue and not white. The reason for this is that the colourless/white copper sulphate absorbs water from the air and this turns it blue. Copper sulphate has a giant ionic lattice structure. The water it absorbs from the air fits into this lattice structure and is held weakly in place. This water can be easily evaporated from the lattice by heating. The addition of water again forms the hydrated blue form of copper sulphate.
When hydrated copper sulphate (blue crystals) are heated in a sealed test tube it undergoes a reversible thermal reaction.
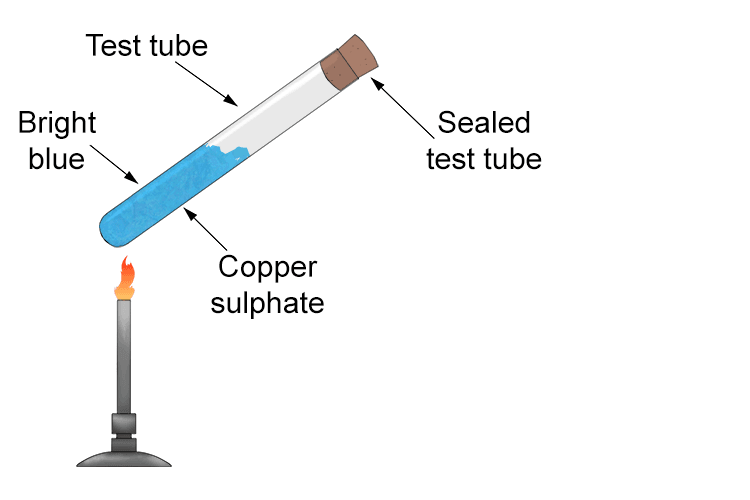
The blue crystals gradually turn into a white powder. The chemical equation is as follows:
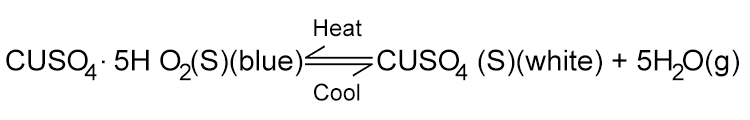
Because the test tube is sealed, the water vapour produced is contained within the tube. The reaction reaches a state of dynamic equilibrium where the forward reaction (hydrate to anhydrous) and the reverse reaction (anhydrous to hydrated) occur at the same rate due to the presence of both the solid reactants/products and the water vapour.
The reaction is reversible. If the heated white powder is allowed to cool it will absorb the water back, reform the blue crystals and the test tube will feel warm (exothermic process).
Another common example is the reaction between nitrogen and hydrogen to make ammonia in the Haber process, where ammonia can also break down back into nitrogen and hydrogen.





