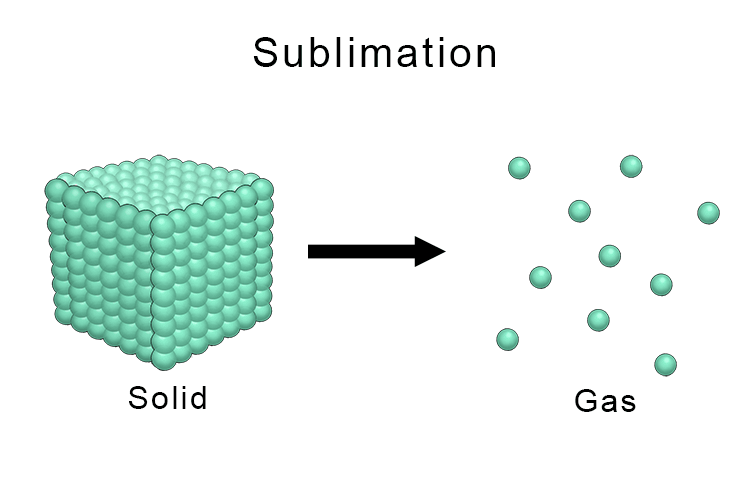
sublimation – process where a substance changes from a solid to a gas directly
(pronounced sub-lih-may-shn)
To remember the meaning of sublimation, use the following mnemonic:
The submarine had a limitation (sublimation), after being built it would turn from solid to gas.

Sublimation is the process where a substance changes directly from a solid state to a gas state without passing through the liquid state. This occurs when particles in a solid gains enough energy to break free from their fixed positions and enter the gas phase, bypassing the melting stage entirely. A common example of sublimation is solid carbon dioxide, also known as dry ice, which turns directly into carbon dioxide gas at room temperature and pressure without becoming liquid first.
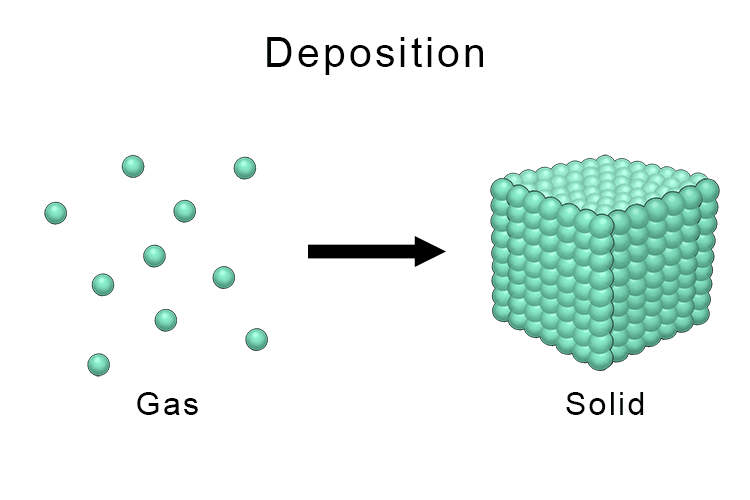
The opposite to sublimation is deposition. Deposition is where a substance changes from a gas to a solid skipping the liquid phase.
An example of deposition in chemistry is the formation of frost on a window where water vapor (gas) in the air changes directly into ice crystals (solid) on the cold glass.