acid rain – rain that is acidic due to dissolved gases (sulfuric acid and nitric acid)
The term acid rain is self explanatory, you can picture acid rain falling on a tree and killing it. Remembering that the primary acids in acid rain are sulfuric acid (H2SO4) and nitric acid (HNO3) is a bit trickier.
To remember that acid rain is sulfuric acid and nitric acid, use the following mnemonic:
The acid rain damaged the sultan's fur (sulfuric acid) coat at night, so he jumped into a rickshaw (nitric acid).

What gases are released when fossil fuels burn:
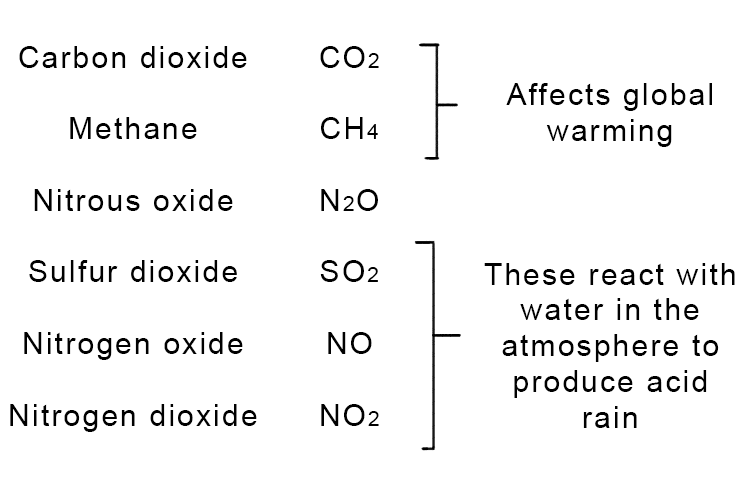
Acid rain = sulfuric acid (H2SO4) and nitric acid (HNO3)
Acid rain is rainfall that has become more acidic that normal due to pollution in the atmosphere. It forms when sulfur dioxide and nitrogen oxides are released into the air, primarily from burning fossil fuels in power stations, factories and vehicle engines. These gases react with water vapour and oxygen in the atmosphere to form sulfuric acid and nitric acid, which then fall to the ground as acid rain, with a pH lower than normal rain (which has a pH of around 5.6). Acid rain has several harmful environmental effects: it damages trees and plants by affecting the nutrients in soil, kills aquatic life in lakes and rivers by making the water too acidic, corrodes buildings and statues, and can harm ecosystems.




