alloy – a mixture of two or more elements, at least one of which is a metal
(pronounced a-loy)
To remember the meaning of alloy, use the following mnemonic:
A loyal (alloy) pair of metal elements were joined together.
An alloy is a mixture of two or more metals, or a metal combined with another element, that is created to improve the properties of the originals pure metal. Pure metals often have limitations such as being too soft, too reactive, or not strong enough for certain applications, so adding another element can enhance their characteristics. For example, steel is an alloy made by mixing iron with small amounts of carbon, which makes it much harder and stronger than pure iron. Similarly, bronze is an alloy of copper and tin, this is harder and more durable than pure copper.
To form an alloy, the base metal is heated until melting point, and as a liquid, the alloying element is then added, forming strong bonds with the alloy when mixed.
This changes the atomic structure of the metals, disrupting the regular order or atoms in a lattice.
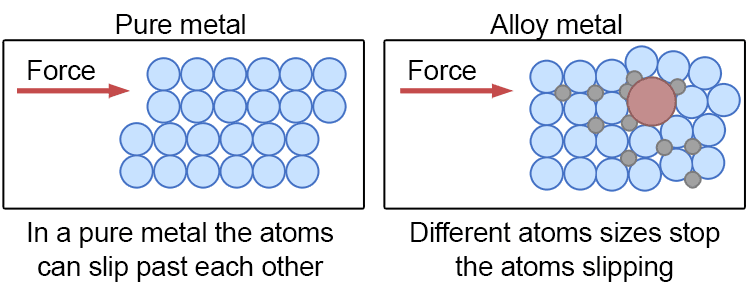
Alloys are important in everyday life because they allow us to create materials with specific desirable properties, such as increased strength, resistance to corrosion, or improved flexibility, making them suitable for a wide range of uses from construction and manufacturing to jewellery and electronics.
Alloys
- Steel - iron and carbon.
- Stainless steel - iron, carbon, chromium and nickel.
- Cast iron - iron and carbon (higher levels than steel).
- Brass - copper and zinc.
- Bronze - copper and tin.
- Solder - tin and lead.
- Pewter - tin, copper, antimony and lead.




