polar molecules – chemical compounds with uneven electron distribution
To remember the meaning of polar molecules, use the following mnemonic:
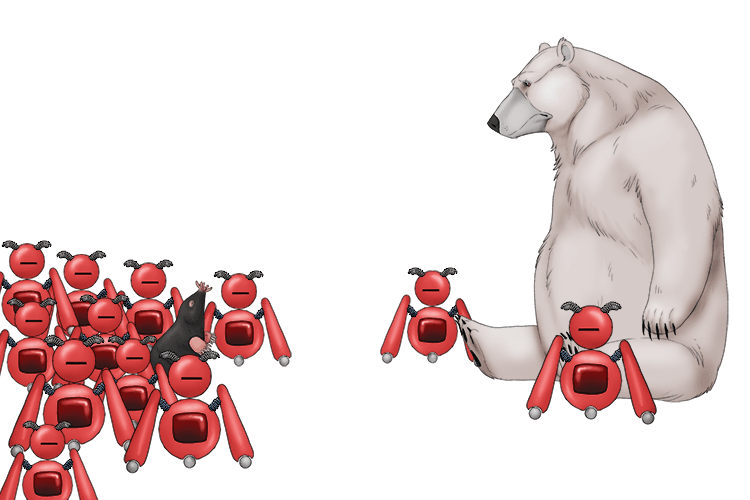
The polar bear and the mole (polar molecule) had an uneven distribution of electronic robots (electron) between them.
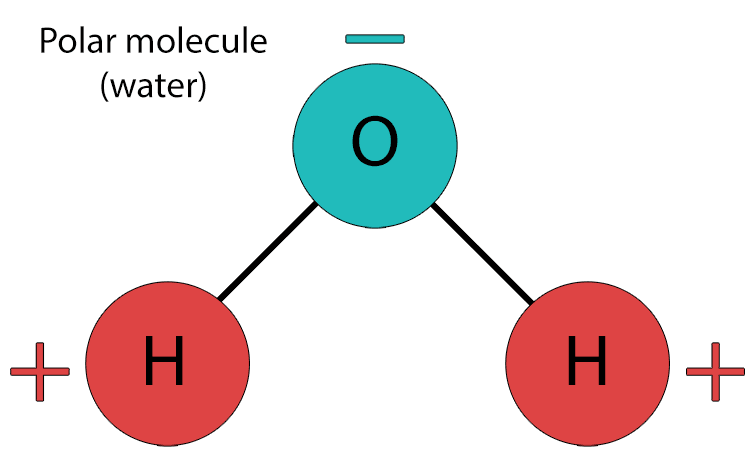
A polar molecule is a molecule that has an uneven distribution of electrons and therefore electrical charge, resulting in one end being slightly positive and the other end being slightly negative. This occurs when there is a difference in electronegativity between atoms in the molecule. Water is the most common example of a polar molecule - oxygen pulls shared electrons closer to itself, creating a slightly negative charge on the oxygen end and slightly positive charges on the hydrogen end.
The opposite is a non-polar molecule, which has an even distribution of electrons and therefore no distinct positive or negative poles. Non-polar and polar molecules have different properties and characteristics. For example, polar molecules are able to dissolve other polar substances (like salt dissolving in water) and have high boiling points due to strong intermolecular forces between molecules. Non-polar molecules will only dissolve in other non-polar molecules and will not mix with polar molecules.
How can you tell if a molecule will be a polar molecule or a non-polar molecule?
Ask yourself two questions:
1. If there are two different terminal atoms around the central atom it is a polar molecule.
2. If the central atom has any lone pairs of electrons it is a polar molecule.
Example NH3 (Ammonia)
Draw a dot and cross diagram.
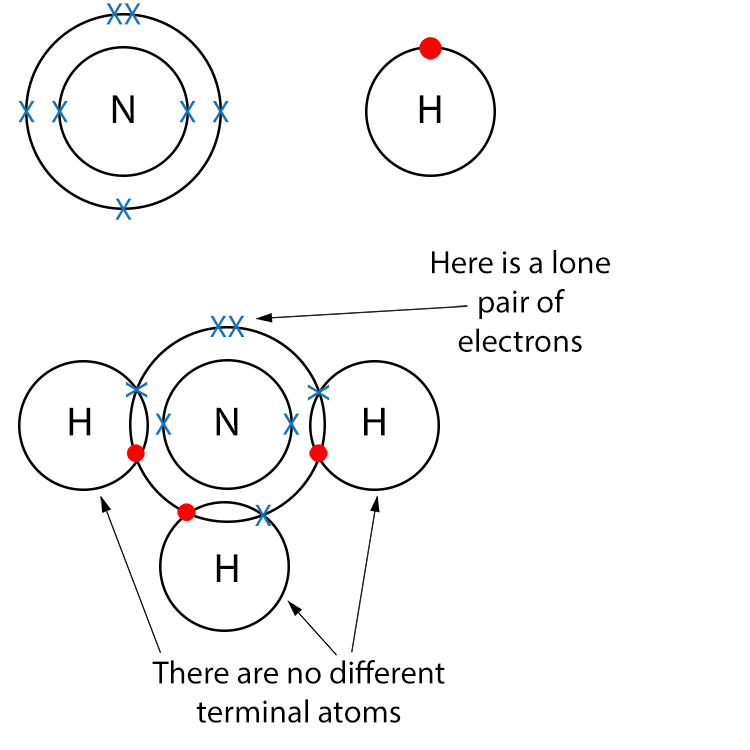
Here we do not have different terminal atoms but we do have a lone pair of electrons so NH3 is a polar molecule.
Example SF6 (Sulfur Hexafluoride)
Draw a dot and cross diagram.
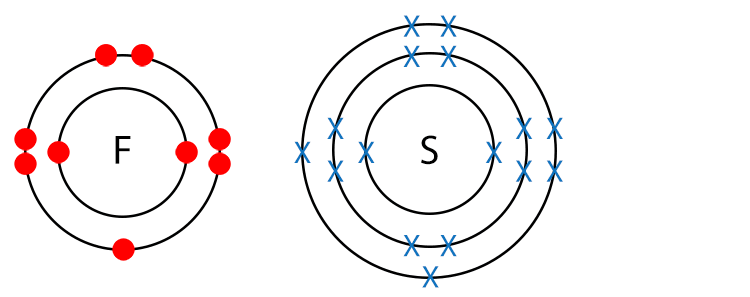
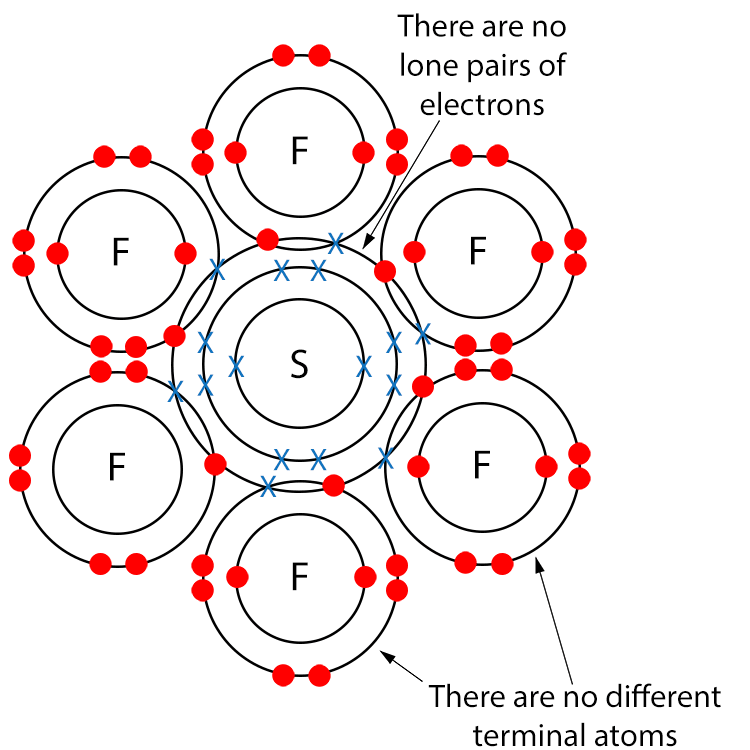
Here we have no different terminal atoms and there are no lone pairs of electrons so sulfur hexafluoride is a non-polar molecule.
Example CCL4 (Carbon Tetrachloride)
Draw a dot and cross diagram.
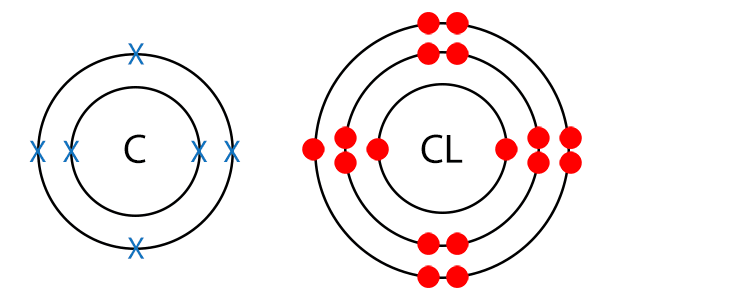
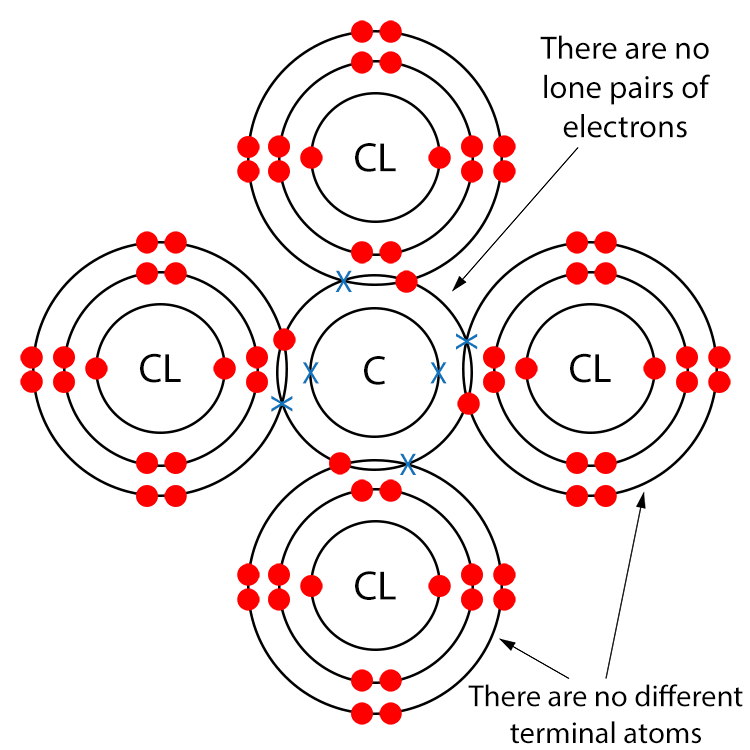
There are no different terminal atoms and there are no lone pairs of electrons, so carbon tetrachloride is a non-polar molecule.
Example CO2 (Carbon Dioxide)
Draw a dot and cross diagram.
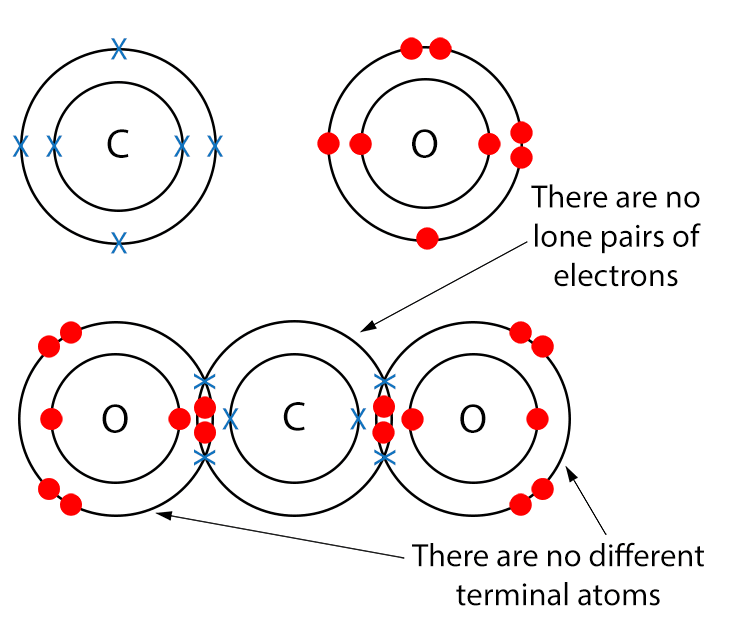
Here we have no different terminal atoms and there are no lone pairs of electrons, so carbon dioxide is a non-polar molecule.
Example H2O (Water)
Draw a dot and cross diagram.
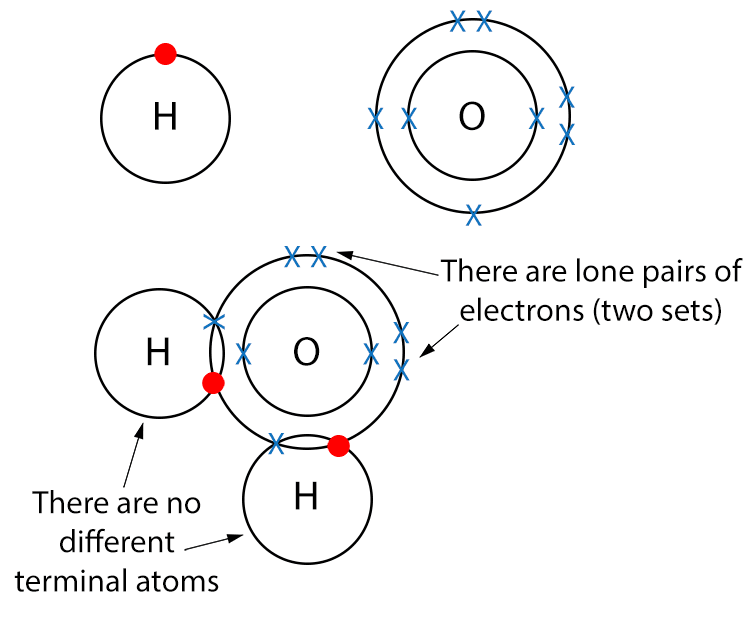
In water molecules there are no different terminal atoms but there are lone pairs of electrons so water is a polar molecule.
Example CH2CL (Chloromethane)
Draw a dot and cross diagram.
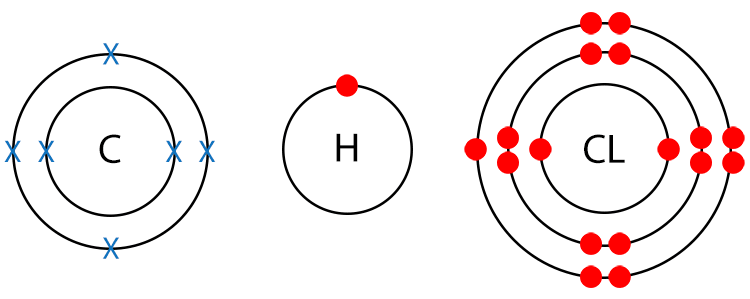
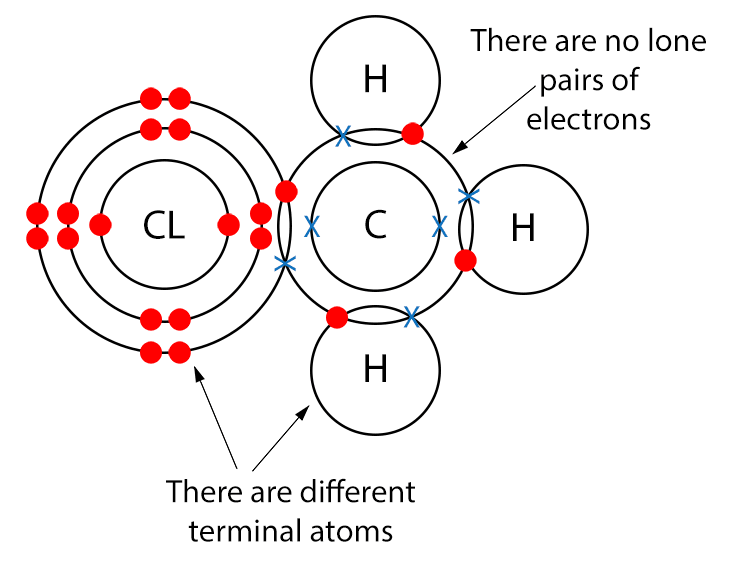
With chloromethane there are no lone pairs of electrons. There are, however, different terminal atoms, so chloromethane is a polar molecule.




