reduction – an atom gains an electron in a chemical reaction (sometimes oxygen is lost)
(pronounced ruh-duk-shn)
Like oxidation the best way to remember the meaning of reduction is to use the following mnemonic:
Think OILRIG. See an oil rig covered with electrons who arrive and leave the oil rig to keep it going.

OILRIG stands for:
OIL - Oxidation Is the Loss of electron.
RIG - Reduction Is the Gain of electron.
When a substance (like iron) reacts with oxygen, iron loses electrons and is oxidised, while oxygen gains electrons and is reduced. The gain of electrons by oxygen is the definition of reduction. Oxidation and reduction occur at the same time.
Example 1
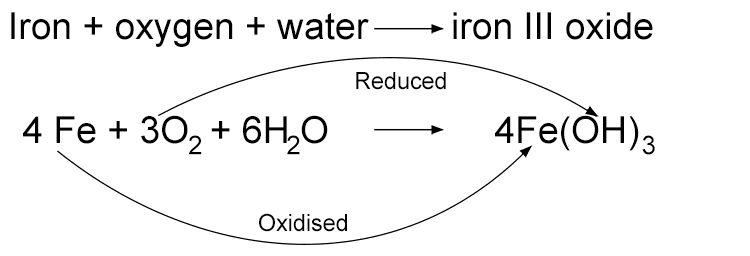
When iron rusts, the oxygen gains electrons from the iron. Along with water the oxygen also forms hydroxide ions (OH–). Because oxygen gains electrons the oxygen is said to have been reduced.
Rusting is a redox reaction, i.e. there is a transfer of an electron.
Reduction does not just occur when oxygen gains electrons. Reduction is now defined as a gain of electrons which occurs in other reactions.
Example 2
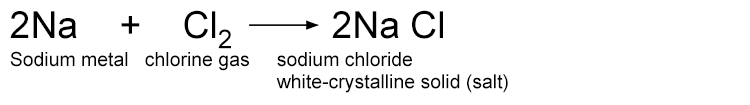
When sodium metal and chlorine gas combine to form salt, the sodium atoms lose electrons to form a sodium ion Na+. Each chlorine atom gains one electron to become a chlorine ion Cl–. The positively charged sodium ion (Na+) and the negatively charged chloride ion (Cl–) are then attracted to each other forming the ionic compound sodium chloride NaCl (salt). Sodium is oxidised, chlorine is reduced.
For a more detailed analysis of ionic bonding, redox reactions, oxidation and reduction see the Mammoth Memory section on chemical bonding.
Example 3
Reduction refers to a chemical reaction where a substance gains electrons. Oxidation always happens alongside reduction. Both are known as redox reactions, where one substance is reduced while the other is oxidised.
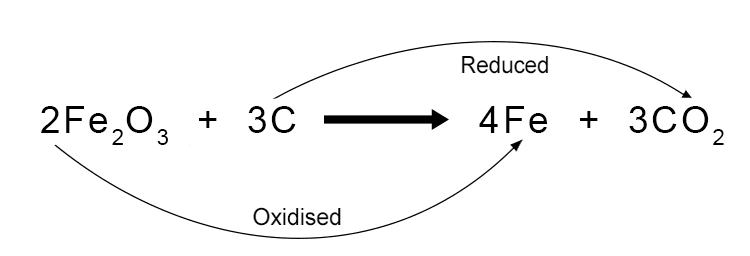
For example, when iron oxide (rust) is heated with carbon to extract iron metal, the iron oxide is reduced because it loses oxygen atoms, while the carbon is oxidised as it gains oxygen to form carbon dioxide. The equation for this reduction reaction is: