state symbols – the symbols used in chemical equations to denote the states of the chemical
(Note: The symbols and states are as follows: (s) - solid, (l) - liquid, (g) - gas, (aq) - aqueous solution).
To remember the meaning of state symbols, use the following mnemonic:
The state police held up symbols (state symbols) to show the different forms of matter.
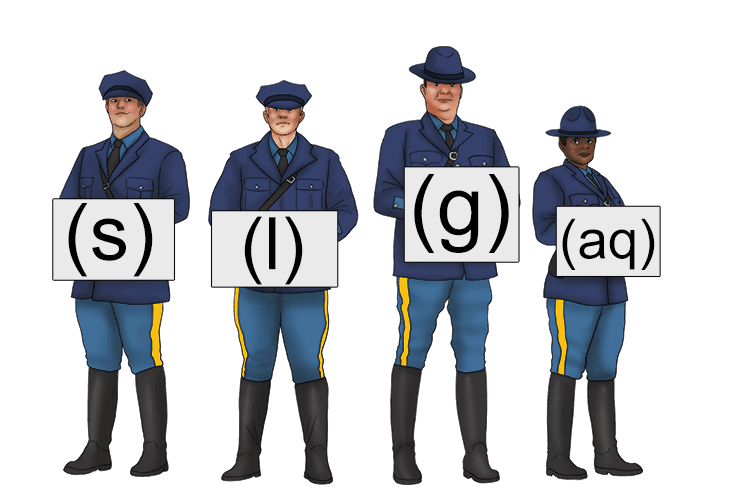
State symbols are abbreviations used in chemical equations to show the physical state of each substance involved in a reaction.
There are four main symbols: (s) for solid, (l) for liquid, (g) for gas, and (aq) for aqueous, which means a substance dissolved in water. These symbols are written in brackets immediately after the chemical formula of each substance in an equation. For example, in the equation NaCl(s) + H2O(l) → NaCl(aq), the state symbols show that solid sodium chloride is mixed with liquid water to produce sodium chloride dissolved in water (salt water). State symbols are important because they provide additional information about the components and results of a chemical reaction.




