haber process – the industrial chemical method for manufacturing ammonia
(pronounced har-buh)
To remember the meaning of Haber process, use the following mnemonic:
At the harbour, processing (Haber process) took so long that it made a crowd moan near (made ammonia) the exit.

The Haber process is used to manufacture ammonia from nitrogen and hydrogen gases. This reversable reaction takes place at high temperatures (around 450°c), high pressures (approximately 200 atmospheres), and uses an iron catalyst to speed up the reaction without being used up itself. The nitrogen is typically extracted from the air, while the hydrogen is often obtained from natural gas or other hydrocarbons. The ammonia produced is extremely important because it's used primarily in fertilisers.
The Haber process demonstrates key concepts in chemical equilibrium, as the reaction is reversible and the conditions must be carefully balanced to achieve a reasonable yield of ammonia while keeping production costs manageable.
A simplified diagram of how the Haber process works is as follows:
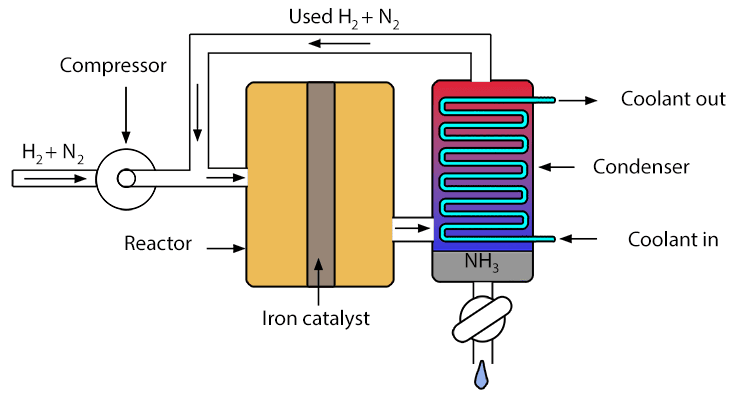
The chemical equation for the Haber process is:
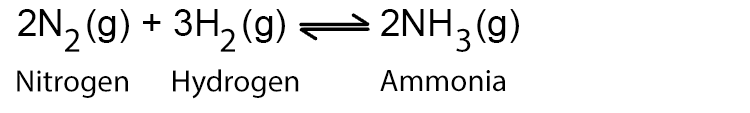
Note: In the Haber process, ammonia is stored by first cooling the mixture of gasses from the reactor, which causes the ammonia to condense into a liquid. The liquid ammonia is then transferred to pressurised storage vessels for storage and transport.
To remember the formula for ammonia, click here.




