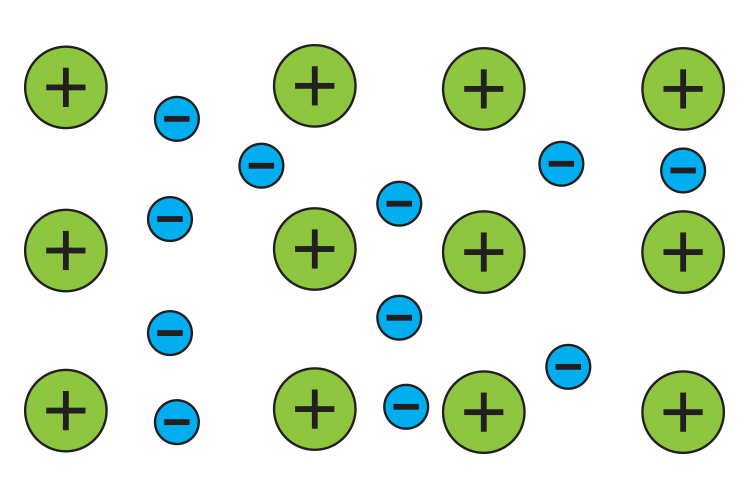
Metals – a sea of electrons
Metals have electrons which do not stay attached to their atoms. Instead these electrons float around inside the metal like a liquid and are often described as a "sea of electrons".
So all wires are full with a sea of electrons and these electrons are moveable which lets metals be electric conductors.
See below.