beta particles – a form of radiation when electrons or positrons are emitted from the nucleus of an atom during radioactive decay
(pronounced bay-tuh)
To remember the meaning of beta particles, use the following mnemonic:
I sat on the bay wall to (beta) watch the electronic robots (electron) be emitted from the night club (electrons emitted).

Beta particles are high-energy, high-speed electrons or positrons (a +ve charged particle) emitted from the nucleus of a radioactive atom during radioactive decay, specifically during a process called beta decay. In the most common form of beta decay (beta-minus decay), a neutron in the nucleus transforms into a proton while emitting a beta particle (an electron) and an antineutrino, increasing the atomic number by one while keeping the mass number the same, effectively changing the element into the next one in the periodic table.
The following examples shows a carbon-14 atom undergoing beta-minus decay to become a nitrogen-14 atom:
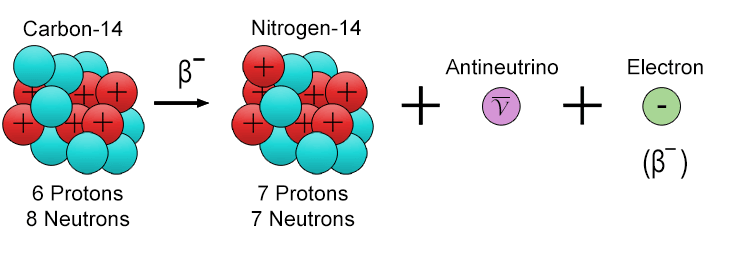
Beta particles are represented by the symbol β⁻ (for electrons) or β⁺ (for positrons in beta-plus decay), and they can travel at speeds approaching the speed of light, typically around 90% of light speed. Compared to alpha particles, beta particles have greater penetrating power (think B for bigger) and can travel several metres in air and penetrate a few millimetres into materials like aluminium. Beta radiation is moderately ionising, meaning it can remove electrons from atoms as it passes through matter, which makes it potentially harmful to living tissue if the source is ingested or inhaled.




