radioactive decay – is a random process in which unstable atomic nucleus loses energy by emitting radiation to become more stable
To remember the meaning of radioactive decay, use the following mnemonic:
After the vet took out the painful radioactive tooth with decay (radioactive decay), the horse calmed down and could go back to the stable (nucleus becomes stable). The energy filled tooth got taken away (energy is emitted) from the room.


Note: A nucleus emits either alpha or beta radiation in a decay. Gamma radiation may also be emitted if the nucleus has excess energy.
Alpha, beta and gamma radiation are all classified as ionising radiation.
Ionisation is the process of knocking electrons out of other atoms or molecules creating charged particles. This can break chemical bonds and damage living tissue which is why ionising radiation is hazardous.
If alpha = 2 protons + 2 neutrons are emitted.
If beta = an electron is emitted.
If gamma = electromagnetic (or energy) is emitted (called photons) which consists of high-energy packets of energy and are massless. Light is electromagnetic and consists of photons.
Note: Random means while the overall decay rate of a large sample is predictable it is impossible to predict any specific individual nucleus will decay.
Measuring Radioactive Decay
When a radioactive atom decays it emits particles of radiation. A Geiger Muller counter is a gas filled tube that conducts electricity when radiation passes through it. When radiation ionises the gas it creates ions that allow a current to flow, producing a measurable pulse.
Rate of Decay
The rate of decay is measured by the time it takes for half of the radioactive material to break down (a half life).
On a Geiger Muller counter this would mean that after one half-life, half the radioactive nuclei remain, so the pulse rate is halved.
Half Lifes.
The decay rate slows down as fewer particles remain. The rate of decay is exponential i.e. after one half-life the pulse rate is halved, after two half-lives it is reduced to a quarter of the original count rate.
This can all be summed up in the following graph:
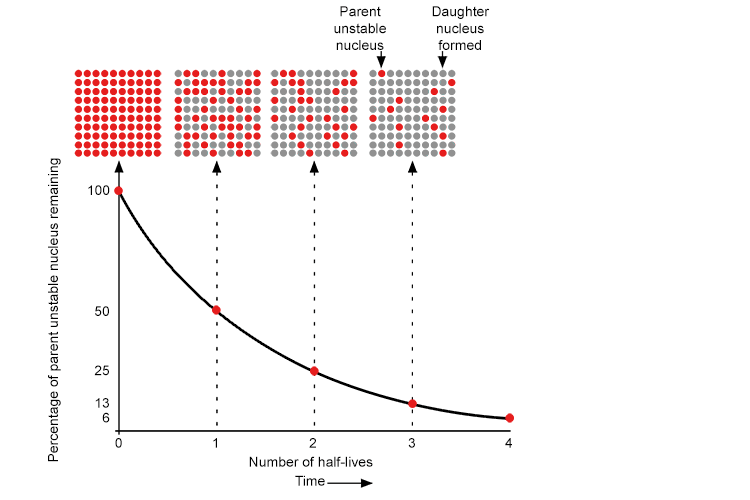
Note: Half-lives are used because it is not dependant on the amount of a radioactive substance.




