density – the measure of how tightly packed molecules are in a substance
(pronounced den-sih-tee)
Note: The formula for density is as follows:
`\text{Density} = (\text{mass})/(\text{volume})`
`\text{Density} = (\text{M})/(\text{V})`
To remember the meaning of density, use the following mnemonic:
The ` forms the top of the heart.
The ` forms the bottom of the heart.
Now you should have a mnemonic that you will never forget.
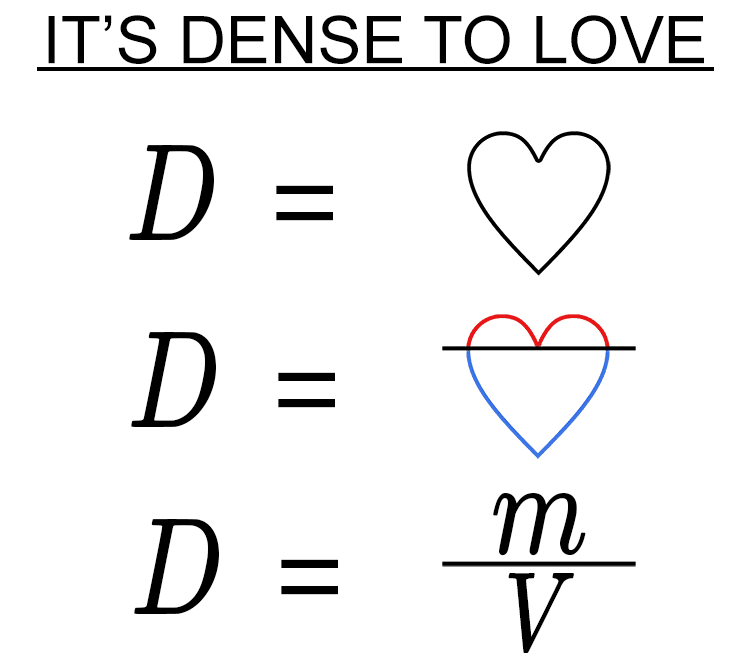
Different materials have different densities because their particles are arranged differently - for example, metals like lead and gold have high densities because their atoms are packed very closely together, while materials like polystyrene or cork have low densities because they contain lots of air spaces.
Density is typically measured in kilograms per cubic metre (kg/m³) or grams per cubic centimetre (g/cm³).
NOTE:
`m =` mass in kilograms (`kg`)
`V =` volume in cubic metres (`m`)
`D =` Density in kilograms per metre cubed (`kg//m^3`)
Example 1
John wants to measure the density of a rock. He finds the volume with the apparatus shown below:

The mass of the rock was measured as being 36g. What is the density of the rock?
Answer:
`Density=(mass)/(volume)`
First convert everything to standard units
Mass of the rock `= 36g = 0.036\ kg`
Volume of rock `=36cm^3-18cm^3=18cm^3=0.000018m^3`
Density of the rock `=0.036/0.000018=2000\ kg//m^3`




