catalyst – substance that makes a reaction speed up
To remember the meaning of catalyst, use the following mnemonic:
The cat listed (catalyst) things that speed up and got a positive reaction from the audience.
A catalyst not only speeds up a reaction but also remains unchanged and is not used up during the process. This means it can be used over and over again to help the same reaction occur multiple times. Catalysts work by lowering the energy required for a chemical reaction to take place, this is known as lowering the activation energy.
In the body, enzymes (biological catalysts) speed up essential reactions such as digestion and metabolism.

The graph below compares the amount of energy required if the process occurred over the same time period, for a reaction with the use of a catalyst or using another method (like heat). You can see using a catalyst lowers the energy required for a reaction (called lowering actuation energy).
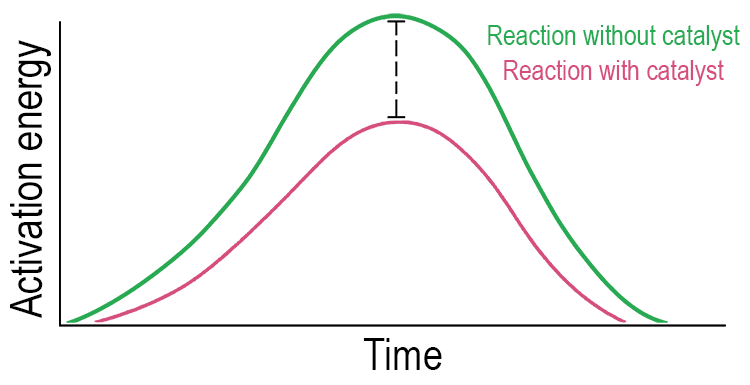
It's important to understand that catalysts only effect the rate of reaction, not the final amount of products formed. They cannot make impossible reactions happen, only ones that would occur naturally, just very slowly.




