osmosis – type of diffusion in which water moves through a semipermeable membrane
(Pronounced oz-moh-sis)
To remember the meaning of osmosis, use the following mnemonic:
The Aussie mows his (osmosis) lawn in the rain (water) and the grass goes from high to low.
Osmosis is a special type of diffusion that involves the movement of water molecules across a selectively permeable membrane from an area of high water concentration to an area of low water concentration. This process occurs when two solutions with different concentrations of dissolved substances are separated by a membrane that allows water to pass through but blocks the movement of larger molecules. Water naturally moves from the dilute solution (which has a higher concentration of water molecules), to the concentrated solution (which has a lower concentration of water molecules) in an attempt to equalise the concentrations on both sides of the membrane.
Osmosis is a passive process, meaning it requires no energy input from the cell and occurs due to the kinetic energy of the water molecules. This process is crucial for many biological functions, including how plant cells maintain pressure to stay rigid, how our kidneys concentrate urine, and how cells regulate their internal water content. In plant cells, osmosis helps maintain cell shape and keeps plants upright, while in animal cells, it's important for maintaining proper cell volume and preventing cells from shrinking or swelling excessively.
Incredibly the force of water wanting to move from a high concentration to a low concentration is so strong that it can more than oppose gravity and cause liquid levels to rise.
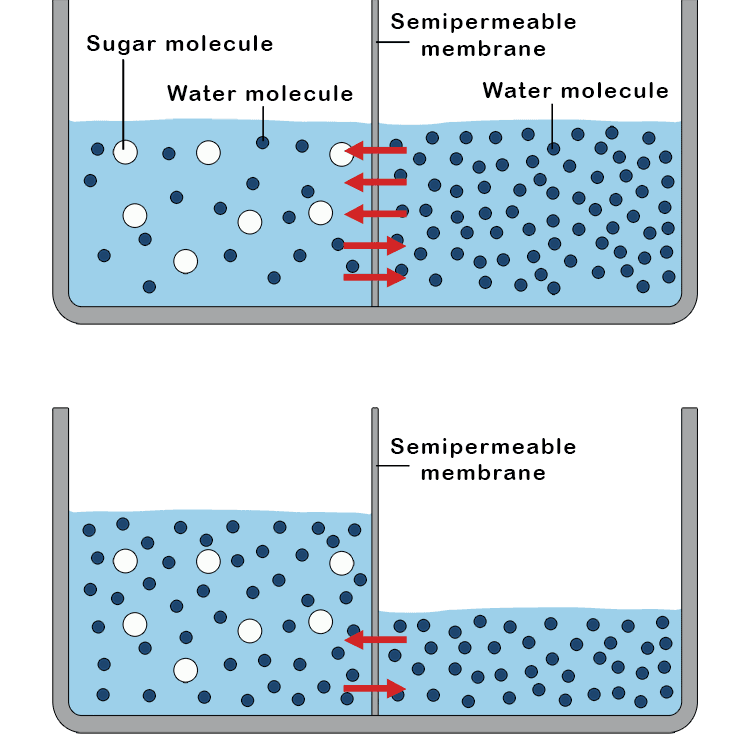
Osmosis causes height differences because it is the net movement of water across a semipermeable membrane from a region of high water concentration (low solute) to a region of low water concentration (high solute). This influx of water into the more concentrated solution compartment increases its volume, causing the liquid level to rise and creating a hydrostatic pressure that opposes further water movement. This difference in height or osmotic pressure, continues until the gravitational force from the taller column balances the osmotic drive for water to move. The increased height of the liquid column creates a pressure (hydrostatic pressure) that pushes water back down, opposing the net flow of water from osmosis.




