advanced electron arrangement – how electrons are distributed in an atom
In early chemistry study, the electron arrangement is discussed as existing in shells of which there is a maximum number of electrons in each shell, 2 electrons in the first, 8 in the second and 8 in the third.
Energy levels
In advanced chemistry, shells are now referred to as energy levels.
In each energy level the number of electrons allowed in each level is worked out with the formula 2 n².
Therefore:
Energy level 1 = 2 X 1² = 2 electrons maximum
Energy level 2 = 2 X 2² = 8 electrons maximum
Energy level 3 = 2 X 3² = 18 electrons maximum
Energy level 4 = 2 X 4² = 32 electrons maximum
We can see the format 2, 8, 8 has now changed to 2, 8, 18, 32.
Note: Importantly energy levels are higher at greater distances from the nucleus (think of it as a higher distance away from the nucleus, a kind of higher potential energy).
Because the attractive force between the positive nucleus and the negative electron weakens as they more further apart.
An electron further away has more total energy. Yes! More.
Sub shells
Electrons are grouped together in sets called sub shells and each has a different shape.
One group is called S
Another P
Another D
Another F
To remember the letters involved in sub shells, use the following mnemonic:
Silly Penguins Dance Frantically

Note: These were originally created to reflect how chemists classified the images of each category they saw i.e. S = Sharp, P = Principal, D = Diffuse and F = Fundamental.
Each sub shell can hold no more than the following number of electrons:
S = 2
P = 6
D = 10
F = 14
The number of electrons in the sub shells follows a pattern of increasing by:
Orbitals
Energy levels contain sub shells and in turn sub shells contain orbitals and orbitals contain up to two electrons.
An orbital is a three-dimensional region of space where an electron is most likely to be found (e.g. 95% probability).
An S orbital is a spherical shaped atomic orbital that can hold a maximum of two electrons.
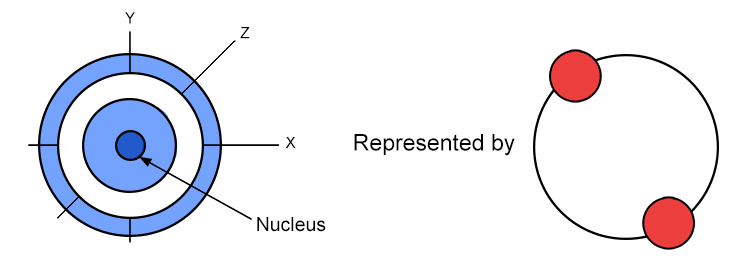
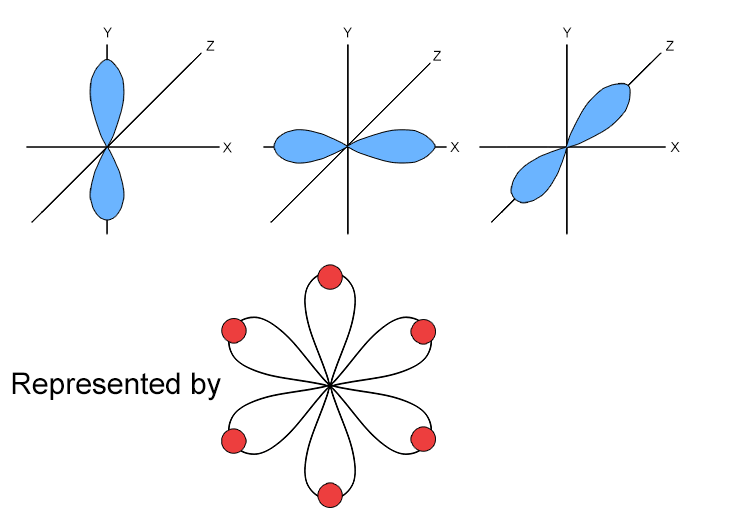
A P orbital is a dumbbell shaped atomic orbital that can hold a maximum of two electrons and are always found in sets of three, oriented at right angles to each other.
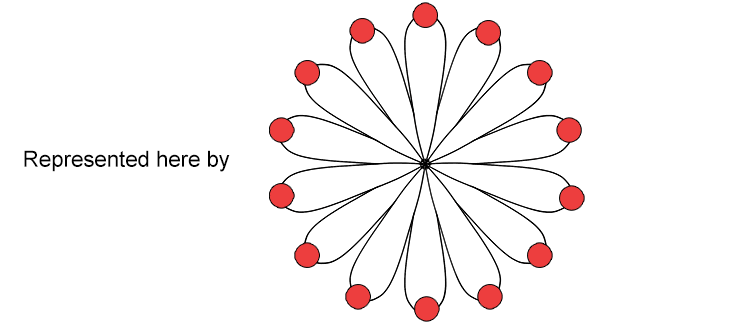
The D orbital can have 5 sets of orbitals, four of which are clover-shaped and one with an elongated dumbbell and doughnut shape.
The F orbital has seven sets of orbitals with complicated shapes.