Be careful with moles
Be careful with mole questions because you need to clearly understand that although all moles of an ideal gas have a volume of 24 litres, they weigh very different amounts.
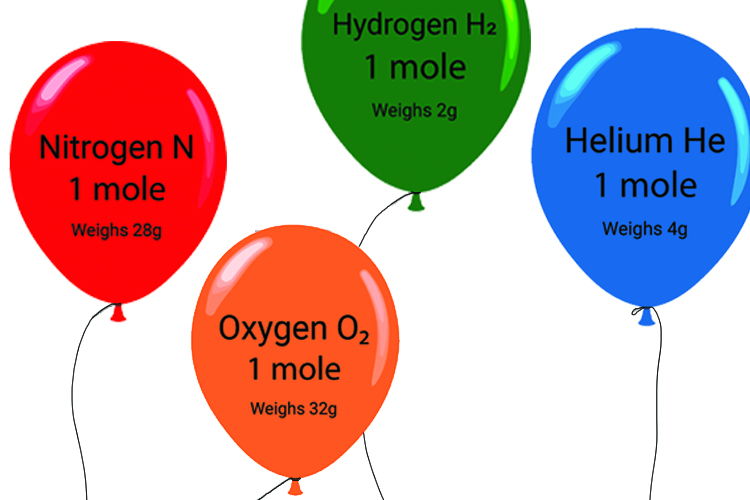
Note:
You can see from the above that even though helium, as an ideal gas, is atomic (1 atom) and hydrogen, as an ideal gas, is diatomic (2 atoms), hydrogen is still half the weight of helium.
In the Zeppelin balloons, in the 1930s, hydrogen was used (not helium).





