Atomic number and mass number
The periodic table shows the unique symbol of every element, together with its mass number and atomic number. For example, lithium (Li) has a mass number of 7 and an atomic number of 3
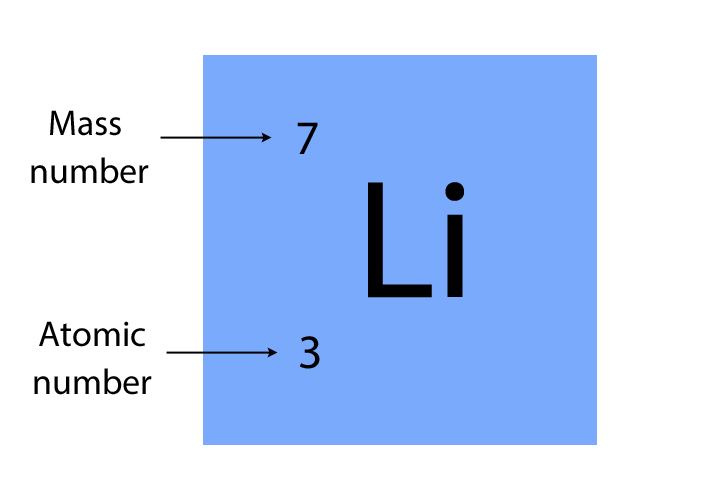
The picture below will help you to remember which number is which.





