Distillation
Distillation is the separation of a solution by vaporising and condensing.
There are two ways you can remember this:
1) Still is in the name 'distillation' (THIS STILL) and a still is what they use in distillation.
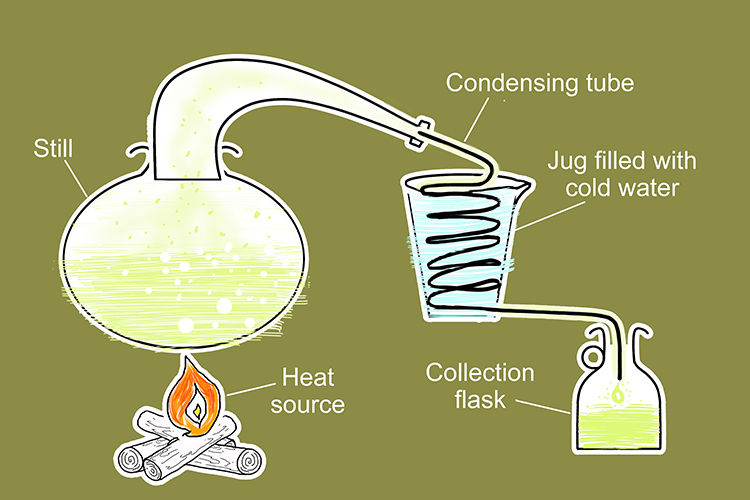 The shape of the still creates a large surface area for the solution which maximises vaporisation.
The shape of the still creates a large surface area for the solution which maximises vaporisation.
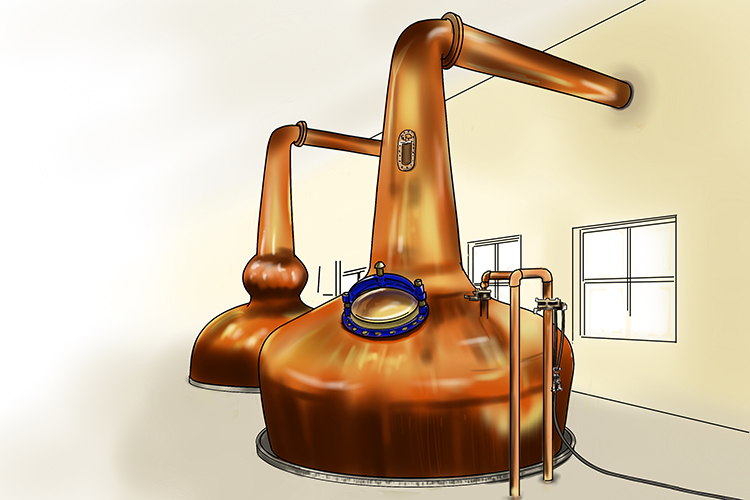 Stills are also used in alcohol distillation plants (they’re usually made of copper).
Stills are also used in alcohol distillation plants (they’re usually made of copper).
2) Separation of a mixed solution by vaporising and condensing.
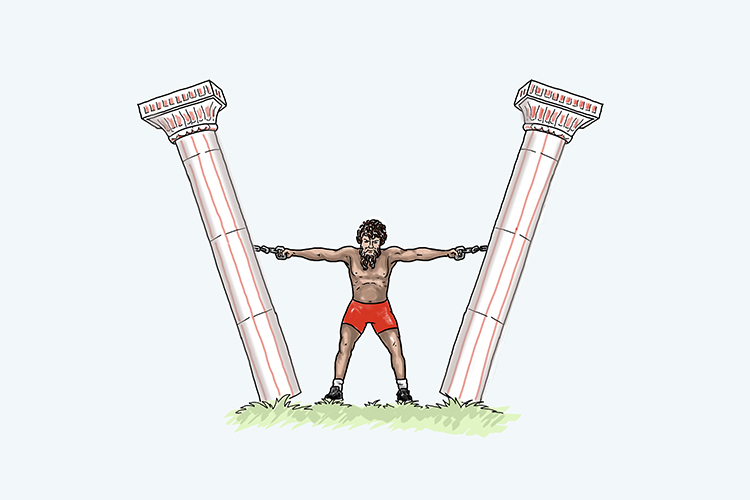 An easy way to remember this is to picture two columns being held together. While they are held, everything remains this still (distill).
An easy way to remember this is to picture two columns being held together. While they are held, everything remains this still (distill).
This is the case until heat is applied, which causes everything to separate (separation).
 Think of an alien vaporising the solution, causing the columns to separate.
Think of an alien vaporising the solution, causing the columns to separate.








