Ethane
Eth- → death → shoe → two → 2 carbon atoms
-ane → cane → shingle → single → single carbon bond
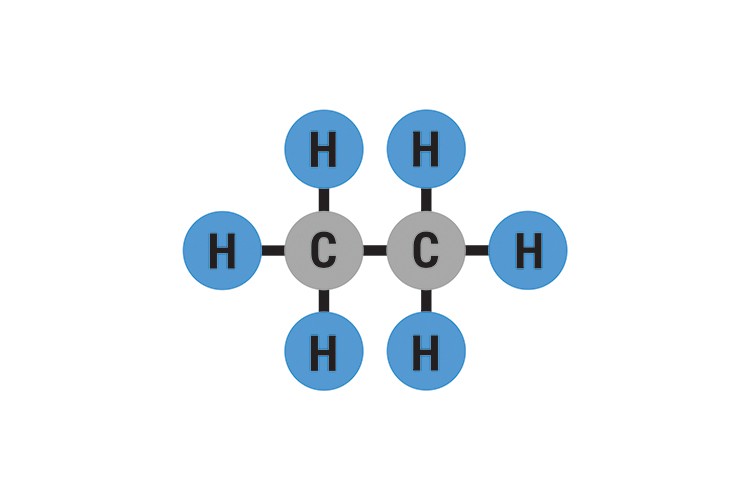
Which looks like
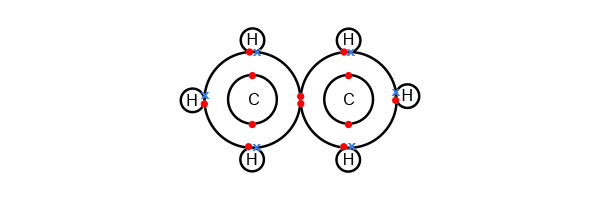
Or
So ethane is C2H6.
C2H6
C2H6 is

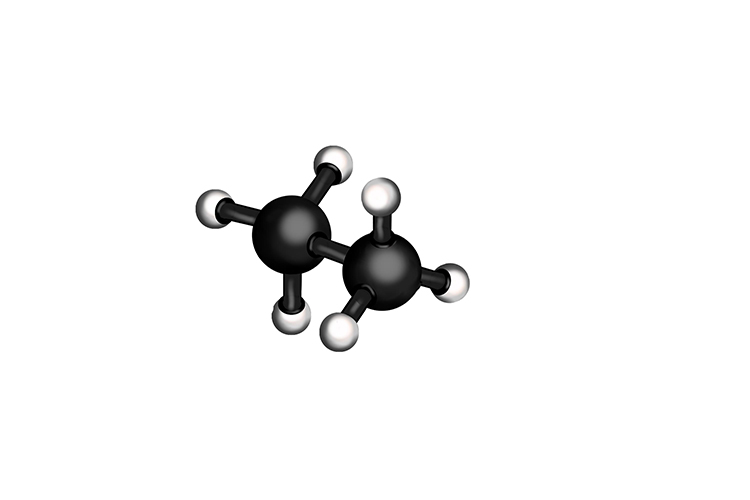
Which looks like

Or

2 carbon atoms → two → shoe → death → eth-
Single carbon bond → single → shingle → cane → -ane
C2H6 = ethane

