Convex lenses and centre of curvature
If you take a clear rectangular block of plastic and cut two circles out of the block as follows:
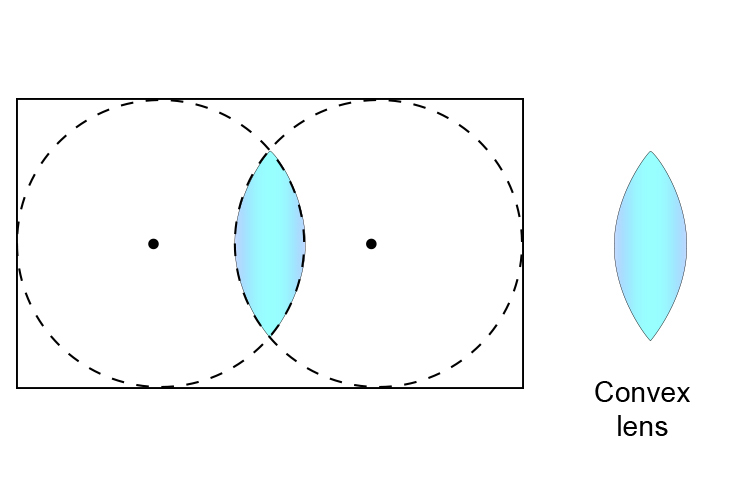
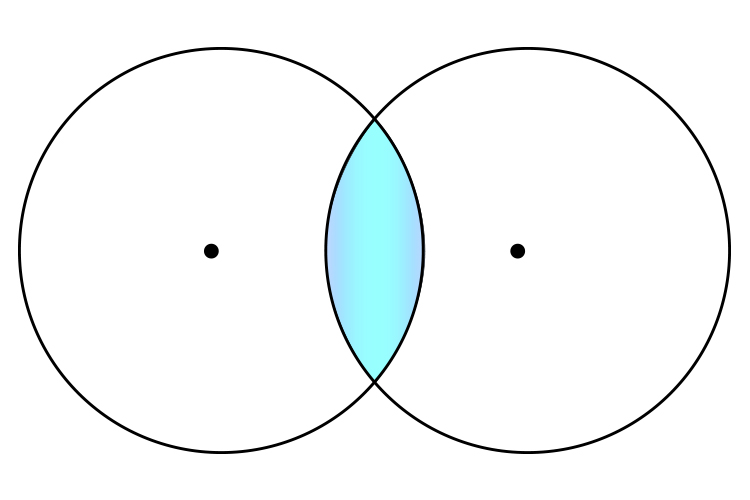
you get a convex lens.
This is great news because this is a terrific way to remember that a convex lens has two centres of curvature (two centres, one in each of two spheres).
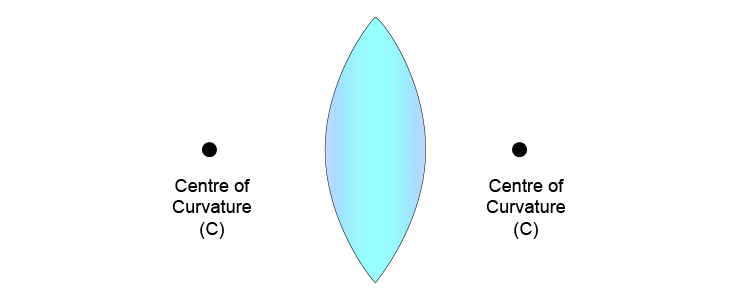
Centres of lens curvatures are usually denoted with a C.




