Electrolysis and water
Chemists always describe the water in a solution that is having electrolysis performed on it as H+ and OH-:
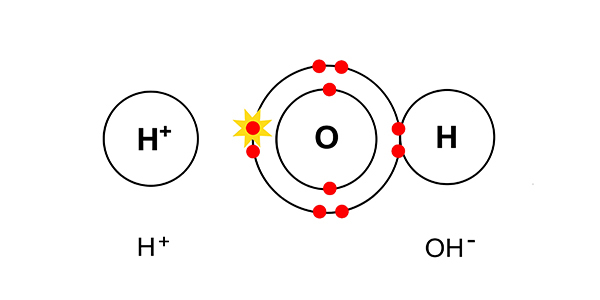
instead of H2O.
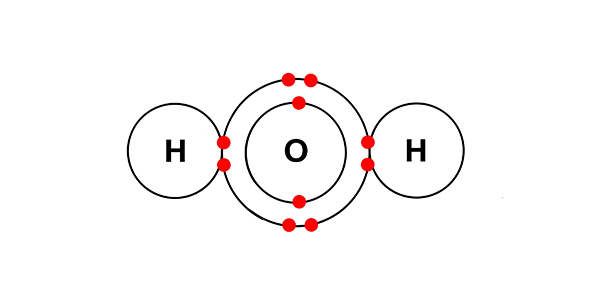





Chemists always describe the water in a solution that is having electrolysis performed on it as H+ and OH-:

instead of H2O.
