How did electrolysis start?
Chemists started putting electrodes – positive and negative electrical terminals connected to a power source via wires – into solutions to see what would happen when they connected a direct-current battery.
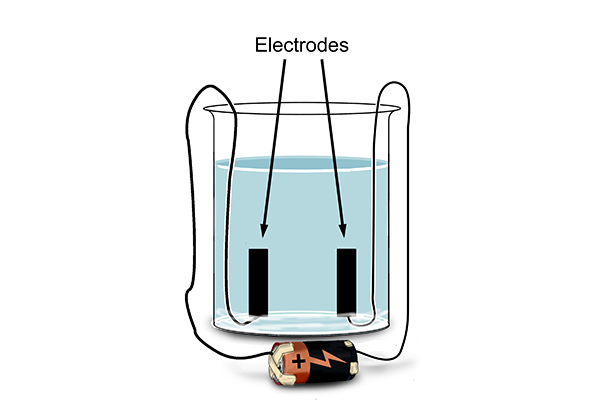
Here are some examples of what happened:
Example 1 – Electrolysis of water and sodium chloride
Example 2 – Electrorefining copper
Example 3 – Electrolysis of water
Example 4 – Electroplating with silver




