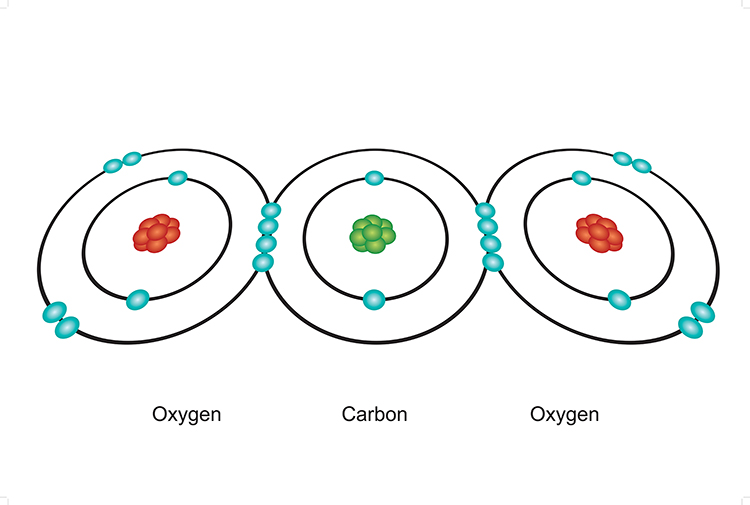
Covalent bonding (sharing)
Covalent bonding is where atoms share electrons.
Think of two people (non-metal) cooperating and covered by a veil (covalent) and deciding to share the rest of their lives together.
Covalent = cooperate = sharing
Covalent = sharing
Example
Within carbon dioxide molecules, a carbon atom shares all of the electrons in its outer shell with two oxygen atoms.